 Image credit: ACS
Image credit: ACSAbstract
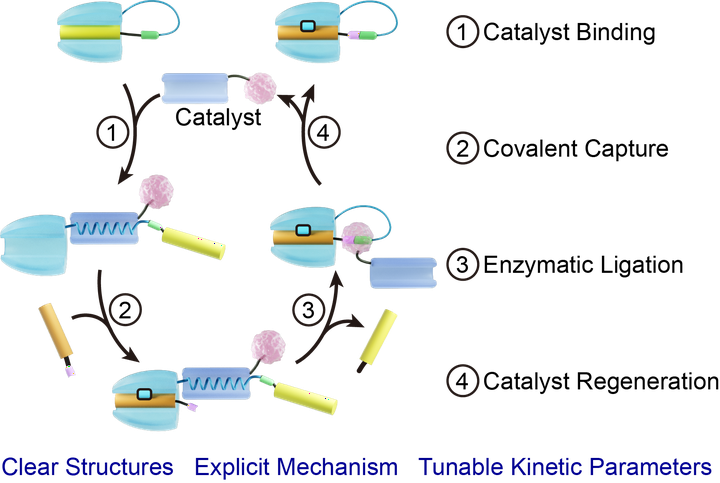
A biological system is rich in dynamic biomolecular assemblyreaction cascades mediated by enzymes and molecular chaperones, as represented by the formation of the “chain-mail-like” bacteriophage HK97 capsid that involves sequential events of chaperone-assisted assembly and cross-linking reactions. To shed light on such catalyzed assembly processes, we report an artificial protein−peptide “assembly-reaction” cascade that can be accelerated by rationally designed catalysts. The cascade is inhibited by a tethered SpyTag mutant that blocks SpyCatcher from the subsequent reactions. A designed fusion of calmodulin and sortase can promote the cascade by first binding with M13 at the loop between the SpyTag mutant and SpyCatcher to open the gating via a coil−helix transition. After the SpyTag-SpyCatcher reaction, the catalyst is regenerated by sortase-mediated cyclization that restores the constrained M13 conformation at the loop to release the bounded calmodulin. In the presence of 0.1 equiv of catalyst, the process can be accelerated, increasing the initial rate by ∼12-fold and reducing the half-life by ∼17-fold. With experimentally measured kinetic parameters, we simulated this system through microkinetic modeling, illustrated the contributions of each parameter, and proposed conditions for optimal catalytic performance. As a prototype of artificial catalyzed supramolecular “assembly-reaction” cascades, this work is reminiscent of those catalyzed cascades in nature. Their common features reveal similar underlying physiochemical principles and suggest new avenues to understand and interfere with biological systems.