Rational design of rigid mRNA folding architecture to enhance intracellular processing and protein production
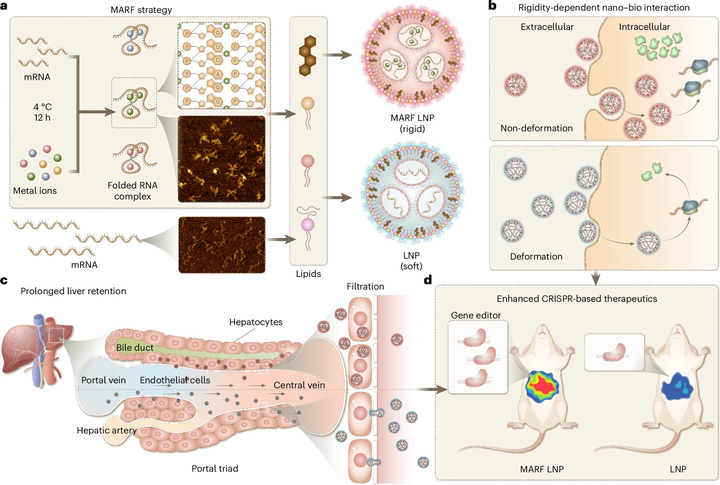 Image credit: Source
Image credit: Source摘要
The application of messenger RNA (mRNA) beyond infectious diseases is challenged by inefficient protein production. Whereas the engineering of secondary mRNA structures has been shown to increase mRNA half-life, it remains unclear whether tertiary mRNA structures influence therapeutic efficacy. Here we develop a metal-ion-assisted RNA folding (MARF) strategy and show that, when delivered with lipid nanoparticles (LNPs), specific metals promote mRNA folding architectures that result in the amplification of protein expression by up to 7.3-fold compared with control mRNA. This effect is due to altered mechanical interactions between the mRNA LNPs and the surrounding biosystem, resulting in enhanced intracellular processing and prolonged retention of delivered mRNA in targeted cells. Administered intravenously, MARF LNPs achieved effective and durable genome editing of the clinically relevant Pcsk9 gene through treatment with a single dose. Overall, this work provides a new MARF technology for more effective mRNA therapy and highlights the potential of mechanical cues in designing nanoparticles for improved mRNA delivery. A metal-ion-assisted RNA folding technique is used to modulate the mechanical interactions between RNA nanoparticles and cell membranes for improved protein expression and mRNA therapy.