Mining 2:2 Complexes from 1:1 Stoichiometry: Formation of Cucurbit[8]uril–Diarylviologen Quaternary Complexes Favored by Electron-Donating Substituents
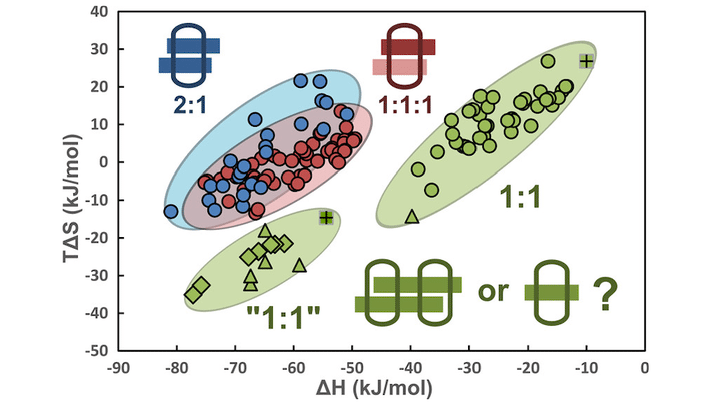 Image credit: ACS
Image credit: ACS摘要
A 1:1 binding stoichiometry of a host–guest complex need not consist of a single host and guest. Diarylviologens containing electron-donating substituents complexed with cucurbit[8]uril (CB[8]) in a 1:1 stoichiometry exhibit abnormally large binding enthalpies compared to typical enthalpy changes observed for 1:1 binary complexes. Here, several CB[8]-mediated host–guest complexes, which were previously reported as 1:1 binary complexes, are verified to be 2:2 quaternary complexes by a combination of isothermal titration calorimetry, $^1$H, NOESY, and ROESY NMR, and ion mobility mass spectrometry, clearly indicating a binding motif of two partially overlapping diarylviologens held in place with two CB[8] molecules. Formation of 2:2 quaternary complexes is favored by electron-donating substituents, while electron-withdrawing substituents typically result in 1:1 binary complexes. The stacking of two highly conjugated diarylviologens in one quaternary motif affords the complexes enhanced conductance when considered as a single-molecular conductor. Moreover, an additional conducting signal previously observed for this “supramolecular” conductor can be readily understood with our 2:2 complexation model, corresponding to a parallel conductance pathway. Therefore, a 2:2 quaternary complex model grants a greater understanding of such supramolecular complexes, enabling the design of engineered, hierarchical structures and functional materials.